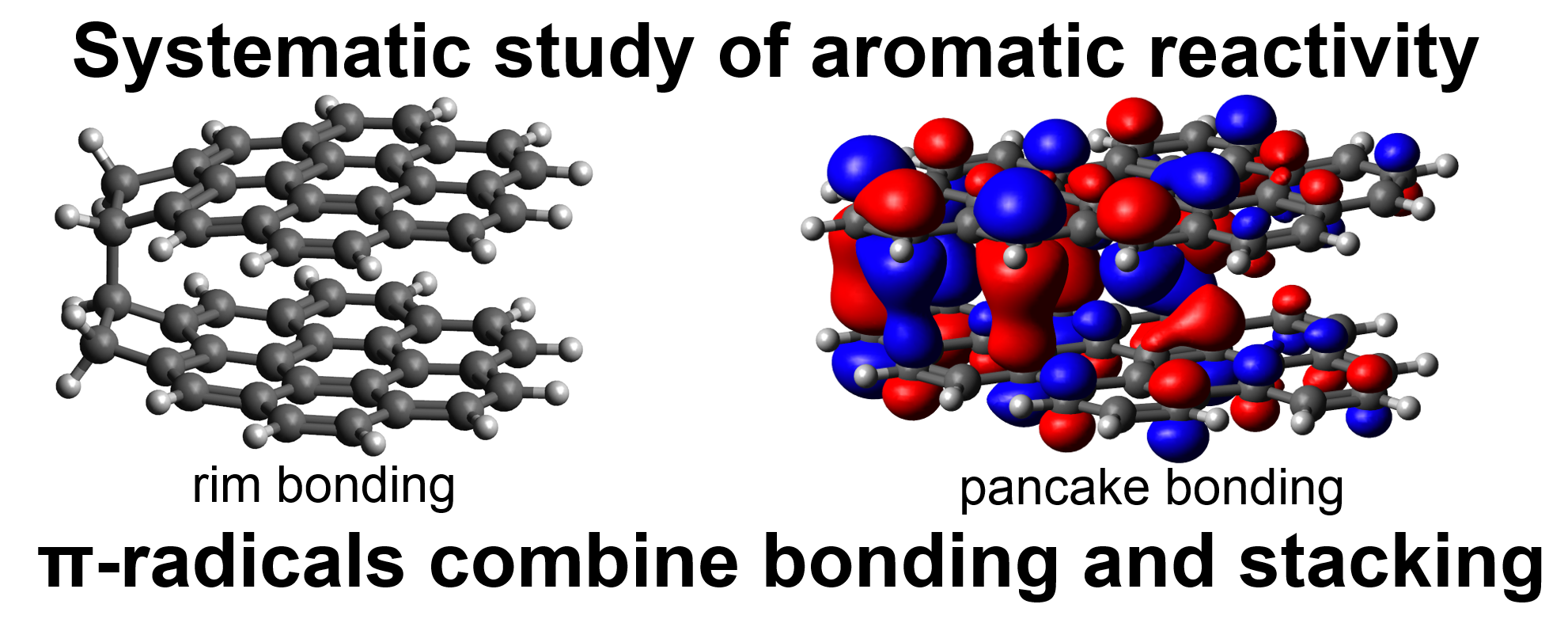
Reactivity of Polycyclic Aromatic Hydrocarbon Soot Precursors: Implications of Localized π-Radicals on Rim-Based Pentagonal Rings
- The reactivity of aromatic soot precursors are studied.
- Covalent bond energies between aromatic soot precursors are calculated.
- Molecular structures combining physical interactions and covalent bonds are shown.
- This paper draws from preprint 234: Reactivity of polycyclic aromatic hydrocarbon radicals: implications for soot formation
- Access the article at the publisher: DOI: 10.1021/acs.jpcc.9b07558