A new methodology to calculate process rates in a kinetic Monte Carlo model of PAH growth
- Development of a combined steady-state and partial-equilibrium approximation.
- Stochastic jump process rate calculation consistent with the chemistry of small gas-phase PAHs.
- Applied in KMC model of PAH formation in premixed flame.
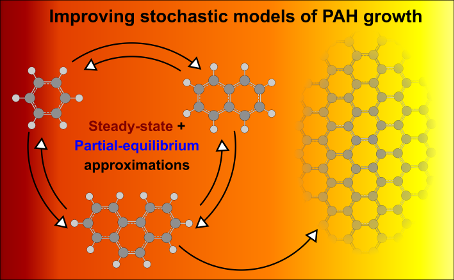 This paper develops a new methodology to calculate the process rates in a kinetic Monte Carlo (KMC) model of polycyclic aromatic hydrocarbon (PAH) growth. The methodology uses a combination of the steady-state and partial-equilibrium approximations. It shows good agreement with the results from simulations using a detailed chemical mechanism under conditions relevant to flames (temperatures between 1000 and 2500 K, equivalence ratios between 0.5 and 10). The new methodology is used to calculate the rate of different stochastic processes in KMC simulations of PAH growth of premixed ethylene-oxygen flames. The resulting rates are only a function of temperature and the main gas-phase species present in the flame environment. The results of the KMC model are shown to be consistent with the concentrations of species calculated using a well-established mechanism for the growth of small PAH species.
This paper develops a new methodology to calculate the process rates in a kinetic Monte Carlo (KMC) model of polycyclic aromatic hydrocarbon (PAH) growth. The methodology uses a combination of the steady-state and partial-equilibrium approximations. It shows good agreement with the results from simulations using a detailed chemical mechanism under conditions relevant to flames (temperatures between 1000 and 2500 K, equivalence ratios between 0.5 and 10). The new methodology is used to calculate the rate of different stochastic processes in KMC simulations of PAH growth of premixed ethylene-oxygen flames. The resulting rates are only a function of temperature and the main gas-phase species present in the flame environment. The results of the KMC model are shown to be consistent with the concentrations of species calculated using a well-established mechanism for the growth of small PAH species.
- This paper draws from preprint 224: A new methodology to calculate process rates in a KMC model of PAH growth
- Access the article at the publisher: DOI: 10.1016/j.combustflame.2019.07.032