Self-assembly of curved aromatic molecules in nanoparticles
- Self-assembly is primarily dependent on cPAH size rather than particle size
- Different cPAH sizes disrupt mesophase formation and arrange in a core-shell particle
- Systems containing cPAHs and fPAHs self-assemble into janus particles
- Cations promote or disrupt particle structure depending on the constituent cPAH size
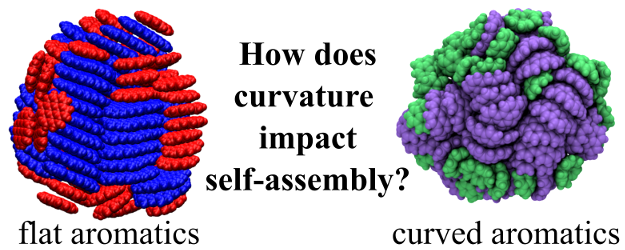 The self-assembly and structure of nanoparticles containing curved polycyclic aromatic hydrocarbon molecules (cPAHs) are investigated using molecular modelling. These polar fullerene-like molecules are receiving increased attention recently due to the steric and electronic properties caused by the inclusion of five-membered ring(s) within their hexagonal lattice. In this work, the curPAHIP potential is extended to describe the interactions between large cPAHs. It is then used within molecular dynamics simulations to produce nanoparticles containing cPAHs. Structural and energetic metrics, including diameter, density, intermolecular spacing, coordination number, alignment angle, radial distance, and energy value, are used to analyse systems containing cPAHs of different sizes and ratios, and containing flat PAHs or ions. Homogeneous cPAH particles are more tightly packed than their flat PAH counterparts, with large cPAHs displaying stacked columnar configurations absent in nanoparticles containing small cPAHs. Mixing cPAHs of different sizes disrupts the ordered mesophase and forms a core-shell structure in which the larger molecules make up the core and the smaller molecules comprise the shell, although this partitioning is less distinct compared to flat PAHs. In addition, the presence of flat PAHs and ions within cPAH nanoparticles promotes distinct arrangements dominated by weak dispersive interactions and strong electrostatic interactions, respectively.
The self-assembly and structure of nanoparticles containing curved polycyclic aromatic hydrocarbon molecules (cPAHs) are investigated using molecular modelling. These polar fullerene-like molecules are receiving increased attention recently due to the steric and electronic properties caused by the inclusion of five-membered ring(s) within their hexagonal lattice. In this work, the curPAHIP potential is extended to describe the interactions between large cPAHs. It is then used within molecular dynamics simulations to produce nanoparticles containing cPAHs. Structural and energetic metrics, including diameter, density, intermolecular spacing, coordination number, alignment angle, radial distance, and energy value, are used to analyse systems containing cPAHs of different sizes and ratios, and containing flat PAHs or ions. Homogeneous cPAH particles are more tightly packed than their flat PAH counterparts, with large cPAHs displaying stacked columnar configurations absent in nanoparticles containing small cPAHs. Mixing cPAHs of different sizes disrupts the ordered mesophase and forms a core-shell structure in which the larger molecules make up the core and the smaller molecules comprise the shell, although this partitioning is less distinct compared to flat PAHs. In addition, the presence of flat PAHs and ions within cPAH nanoparticles promotes distinct arrangements dominated by weak dispersive interactions and strong electrostatic interactions, respectively.
- This paper draws from preprint 267: Self-assembly of curved aromatic molecules in nanoparticles
- Access the article at the publisher: DOI: 10.1016/j.carbon.2021.05.013