Atomic insights into the sintering process of polycyclic aromatic hydrocarbon clusters
- Sintering of homogeneous PAH clusters is studied by means of MD.
- The sintering rate depends on the melting point of the cluster.
- Above the melting point, sintering is rapid and not affected by crosslinks.
- Below the melting point, sintering is slow and hindered by crosslinks.
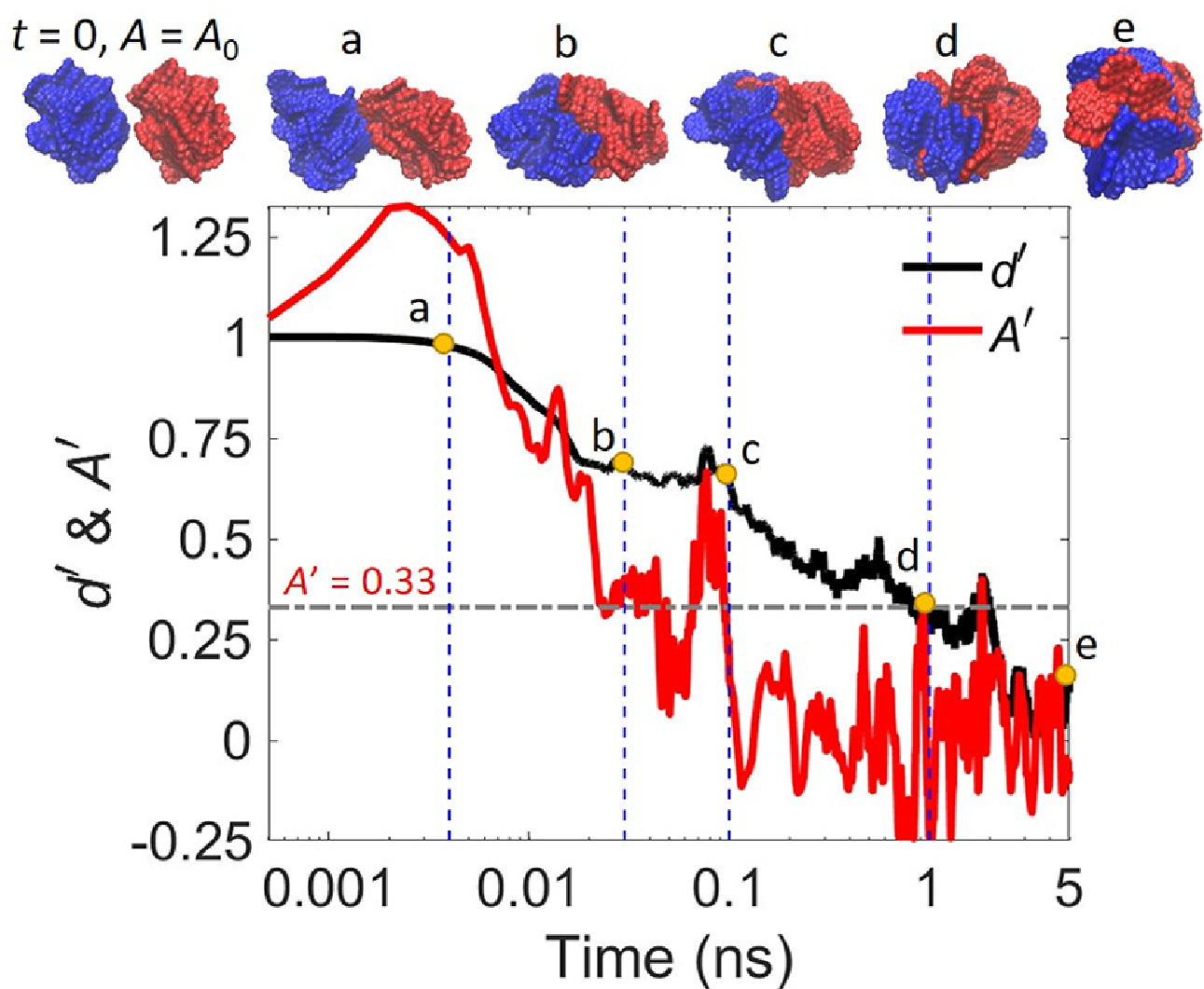 In this work, we studied the sintering process of two homogeneous polycyclic aromatic hydrocarbon (PAH) clusters with diameters in the range of 3–6 nm using molecular dynamics (MD) simulations. The sintering process was quantified through monitoring the solvent accessible surface area (SASA) and the distance between the center of mass (COM) of the two PAH clusters. The effect of temperature and crosslinking level of PAH clusters on sintering was investigated. The results show that the sintering rate of two PAH clusters at a certain temperature T is largely dependent on the melting point (TMP) of the PAH cluster. When T is higher than TMP, the characteristic sintering time (τs) is around 10-2 ns and sintering is not affected by the crosslinking level as the PAH clusters are liquid-like. In contrast, when T is much lower than TMP, the PAH clusters sinter rather slowly with τs > 5 ns, and the sintering process is hindered by the crosslinks between PAH molecules within solid-like PAH clusters due to the enhanced steric effect.
In this work, we studied the sintering process of two homogeneous polycyclic aromatic hydrocarbon (PAH) clusters with diameters in the range of 3–6 nm using molecular dynamics (MD) simulations. The sintering process was quantified through monitoring the solvent accessible surface area (SASA) and the distance between the center of mass (COM) of the two PAH clusters. The effect of temperature and crosslinking level of PAH clusters on sintering was investigated. The results show that the sintering rate of two PAH clusters at a certain temperature T is largely dependent on the melting point (TMP) of the PAH cluster. When T is higher than TMP, the characteristic sintering time (τs) is around 10-2 ns and sintering is not affected by the crosslinking level as the PAH clusters are liquid-like. In contrast, when T is much lower than TMP, the PAH clusters sinter rather slowly with τs > 5 ns, and the sintering process is hindered by the crosslinks between PAH molecules within solid-like PAH clusters due to the enhanced steric effect.
- Access the article at the publisher: DOI: 10.1016/j.proci.2020.06.368